
Alveolar Gas Exchanges
While other parts of the respiratory system move air and conduct
it into and out of the air passages, the alveoli carry on the
vital process of exchanging gases between the air and the blood.
Alveoli
The alveoli are microscopic air sacs clustered at the distal ends
of the finest respiratory tubes, the alveolar ducts. Each alveolus
consists of a tiny space surrounded by a thin wall, which separates
it from adjacent alveoli.
Respiratory Membrane
 |
The wall of an alveolus consists of an inner lining of simple squamous epithelium and a dense network of capillaries, which are also lined with simple squamous epithelial cells. Thin fused basement membranes separate the layers of these flattened cells, and between them there are elastic collagenous fibers, which help to support the wall. There are at least two thicknesses of epithelial cells and a layer of fused basement membranes between the air in an alveolus and the blood in a capillary. These layers make up the respiratory membrane, which is of vital importance because it is through this membrane that gas exchanges occur between the blood and alveolar air. |
Membrane
Gas molecules diffuse from regions where they are in higher concentration toward
regions where they are in lower
concentration. Similarly, gases move from
regions of higher pressure toward regions of lower
pressure, and the pressure of gas
determines the rate at which it will diffuse from one region to
another.
Measured by volume, ordinary air is about 78
percent nitrogen, 21 percent oxygen, and 0.04 percent carbon dioxide.
Air also contains small amounts of other gases that have little or no
physiological importance.
In a mixture of gases, such as air, each gas is
responsible for a portion of the total weight of pressure produced by
the mixture. The amount of pressure each gas creates is called the
partial pressure, and it is directly related to the concentration of the
gas in the mixture. For example, because air is 21 percent oxygen,
this gas is responsible for 21 percent of the atmospheric pressure.
Since 21 percent of 760 mm Hg is equal to 160 mm Hg, it is said that
the partial pressure of oxygen, symbolized Po2, in atmospheric air is 160 mm Hg. Similarly, the partial
pressure of carbon dioxide (Pco2) in air can be calculated as 0.3 mm Hg.

When a mixture of gases dissolves in the blood,
each gas exerts its own partial pressure in proportion to its
dissolved concentration. furthermore, each gas will diffuse between
the blood and its surroundings, and this movement will tend to
equalize its partial pressures in the two regions.
For example, the Pco2 in capillary blood is 45 mm
of Hg, but the Pco2 in alveolar air is 40 mm of Hg. As a consequence of the
difference between these partial pressures, carbon dioxide diffuses
from the blood, where its pressure is higher, through the respiratory
membrane and into the alveolar air. When the blood leaves the lungs,
its Pco2 is
40 mm Hg, which is about the same as the Pco2 of the alveolar air.
Similarly, the Po2 of capillary blood is 40 mm
Hg, but that of alveolar air is 104 mm Hg. Thus, oxygen diffuses from
the alveolar air into the blood, and the blood leaves the lungs with
a Po2 of 104
mm Hg.
Transport Of Gases
The transport of oxygen and carbon dioxide between the lungs and body
cells is a function of the blood. As these gases enter the blood,
they dissolve in the liquid portion (plasma). They combine chemically
with various blood component, and most are carried in combination
with other atoms and molecules.
Oxygen Transport
Almost all of the oxygen (over 98 percent) carried in the blood is
combined with the iron-containing compound, hemoglobin, that occurs within
the red blood cells. The remainder of the oxygen is dissolved in the
blood plasma.
In the lungs, where the Po2 is relatively high, oxygen
dissolves in the blood and combines rapidly with the iron atoms of
hemoglobin. The result of this chemical reaction is a new substance
called oxyhemoglobin.
The chemical bonds that form between the oxygen
and hemoglobin molecules are relatively unstable, and as the
Po2
decreases, oxygen is released from oxyhemoglobin molecules. This
happens in tissues where the cells have used oxygen in their
respiratory processes, and the free oxygen diffuses from the blood
into nearby cells.
The amount of oxygen released from oxyhemoglobin
is affected by several other factors, including the blood
concentration of carbon dioxide, the blood pH, and the blood
temperature. Thus, as the concentration of
carbon dioxide increase, as the blood becomes more acidic, or as the
blood temperature increases, more oxygen is released.
Due to these factors, more oxygen is released to the skeletal
muscles during periods of physical
exercise, because the increased muscular activity accompanied by an
increase use of oxygen causes an increase
in the carbon dioxide concentration, a
decrease in the pH, and a rise in the
temperature. At the same time, less active
cells receive relatively smaller amounts of oxygen.
Carbon Dioxide Transport
Blood flowing through the capillaries of the body tissues gains
carbon dioxide because the tissues have a relatively high
Pco2. This
carbon dioxide is transported to the lungs in one of three forms: as
carbon dioxide dissolved in the blood, as part of a compound formed
by bonding to hemoglobin, or as part of a bicarbonate ion.
The amount of carbon dioxide that dissolves in the
blood is determined by its partial pressure. The higher the Pco
2 of the
tissues, the more carbon dioxide will go into solution. However, only
about 7 percent of the carbon dioxide is transported in this
form.

Unlike oxygen, which combines with the iron atoms
of hemoglobin molecules, carbon dioxide bonds with the amino groups
(-NH2) of
these molecules. Consequently, oxygen and carbon dioxide do not
compete for bonding sites, and both gases can be transported by a
hemoglobin molecule at the same time.
When carbon dioxide combines with hemoglobin, a
loosely bound compound called carbaminohemoglobin is formed.
This substance decomposes readily in regions where the Pco
2 is low
and, thus, releases its carbon dioxide. Although this method of
transporting carbon dioxide is theoretically quite effective,
carbaminohemoglobin forms relatively slowly. It is believed that only
about 23 percent of the total carbon dioxide in the blood is carried
this way.
The most important carbon dioxide transport
mechanism involves the formation of bicarbonate ions (HCO3-). Carbon dioxide reacts with
water to form carbonic acid (H2CO3)
CO 2 + H2O ---> H2CO3
Although this reaction occurs slowly in the blood
plasma, much of the carbon dioxide diffuses into the red blood cells,
and these cells contain an enzyme, called carbonic anhydrase, that speeds
the reaction between carbon dioxide and water.
The resulting carbonic acid then dissociates,
releasing hydrogen ions (H+) and bicarbonate ions
(HCO3-).
H2CO3 -----> H+ + HCO3-
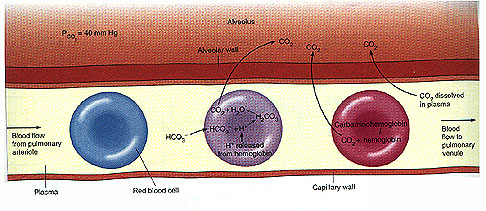
Most of the hydrogen ions combine quickly with
hemoglobin molecules and, thus, are prevented from accumulating and
causing great change in the blood Ph, The bicarbonate ions tend to
diffuse out of red bllod cells and enter the blood plasma. It is
estimated that nearly 70 percent of the carbon dioxde transported in
the blood is carried in this form.
When the blood passes through the capillaries of
the lungs, it loses its dissolved carbon dioxide by diffusion into
the alveoli, This occurs in response to relatively low Pco
2 of the
alveolar air. At the same time, hydrogen ions and bicarbonate ions in
the red blood cells recombine to form carbonic acid molecules, and
under the influence of carbonic anhydrase, the carbonic acid gives
rise to carbon dioxide and water,
H+ + HCO3- ----> CO2 + H2O
Carbaminohemoglobin also releases its carbon
dioxide, and as carbon dioxide continues to diffuse out of the blood,
and equillibrium is established between the Pco2 of the blood and the
Pco2 in the
alveolar air.